
Chemistry, 28.08.2020 20:01 eshaesmot12345
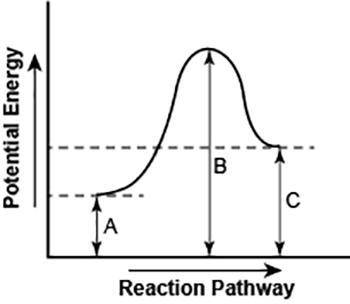
Part 1: Describe how you can determine the total change in enthalpy and activation energy from the diagram, and if each is positive or negative. Part 2: Describe how the curve will look if the reaction was exothermic. Be sure to mention changes in the potential energies of the reactants and products and the sign changes of the enthalpy.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Select the correct answer. you have a nightlight plugged into an outlet in the hallway, which uses 3.5 watts when plugged in. if the house circuit provides 120.0 volts, what is the current through this bulb?
Answers: 1

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 23.06.2019 03:50
Which best describes the activation energy of a chemical reaction? a. the combined energy of all the reactants b. the amount of energy required for a reaction to occur c. the difference in energy between products and reactants d. the potential energy stored in the bonds of reactants and products
Answers: 1
You know the right answer?
Part 1: Describe how you can determine the total change in enthalpy and activation energy from the d...
Questions




Mathematics, 17.01.2020 22:31

Physics, 17.01.2020 22:31

History, 17.01.2020 22:31

Computers and Technology, 17.01.2020 22:31



English, 17.01.2020 22:31

Mathematics, 17.01.2020 22:31


History, 17.01.2020 22:31

History, 17.01.2020 22:31




Mathematics, 17.01.2020 22:31




