
Chemistry, 27.09.2020 20:01 aideramos2855
Question 1
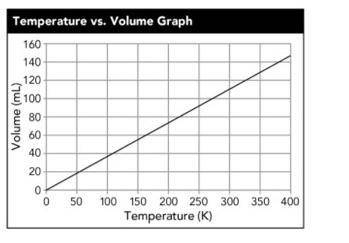
Kenneth warmed up a 100-mL sample of air in an expandable container. He displayed the results in the following graph. The results formed a straight line, so he extended the line the full length of the graph. At what temperature did the gas have a volume of 110 mL?
A. 19 k
B. 300 k
C. 110 k
D. 20 K
Question 2
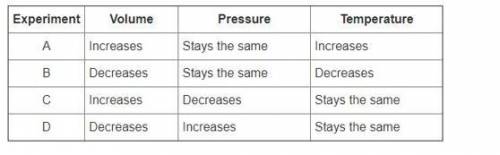
Sonya and Jeremy performed several experiments to show how changes in temperature affected the volume of an inflated balloon. They made sure the pressure stayed the same. They also showed the effects of pressure on volume if temperature stayed the same. They organized their information in the table below.
Which experiments provide evidence for Boyle’s law? Select all that apply.
Experiment A
Experiment B
Experiment C
Experiment D

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1


Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1
You know the right answer?
Question 1
Kenneth warmed up a 100-mL sample of air in an expandable container. He displayed the re...
Questions






Advanced Placement (AP), 08.10.2019 21:30

Mathematics, 08.10.2019 21:30




Biology, 08.10.2019 21:30






Computers and Technology, 08.10.2019 21:30

Mathematics, 08.10.2019 21:30




