You can consult your Periodic Table to find the atomic weights of each element.
Question 5
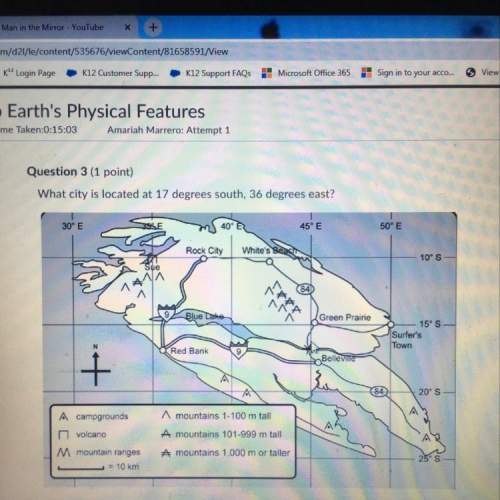
Wh...

You can consult your Periodic Table to find the atomic weights of each element.
Question 5
What is the relative atomic mass of
the element?
Isotope ?-90 has an atomic mass of 90. It has a relative
abundance of 51.5%.
Isotope ?-91 has an atomic mass of 91. It has a relative
abundance of 11.2%.
(Round to the first decimal point)
Submit Answer or Skip Question
Isotope ?-92 has an atomic mass of 92. It has a relative
abundance of 17.1%.
Isotope ?-94 has an atomic mass of 94. It has a relative
abundance of 17.4%.
Isotope ?-96 has an atomic mass of 96. It has a relative
abundance of 2.8%.
Next Chapter: Groups
About this Interactive | Periodic Site Map

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
Questions



Mathematics, 14.08.2021 15:40

English, 14.08.2021 15:40


Social Studies, 14.08.2021 15:40

English, 14.08.2021 15:40





Advanced Placement (AP), 14.08.2021 15:40

Mathematics, 14.08.2021 15:40


Biology, 14.08.2021 15:40

Mathematics, 14.08.2021 15:40

Physics, 14.08.2021 15:40

Mathematics, 14.08.2021 15:40

Mathematics, 14.08.2021 15:40

Social Studies, 14.08.2021 15:40