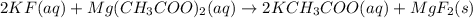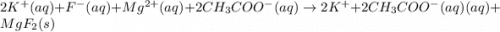Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2


Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
For the following reaction in aqueous solution, identify all the species that will be spectator ions...
Questions



Chemistry, 11.04.2020 01:50

Mathematics, 11.04.2020 01:50

Mathematics, 11.04.2020 01:50

Mathematics, 11.04.2020 01:50

English, 11.04.2020 01:50

Law, 11.04.2020 01:50






Mathematics, 11.04.2020 01:50

Social Studies, 11.04.2020 01:50