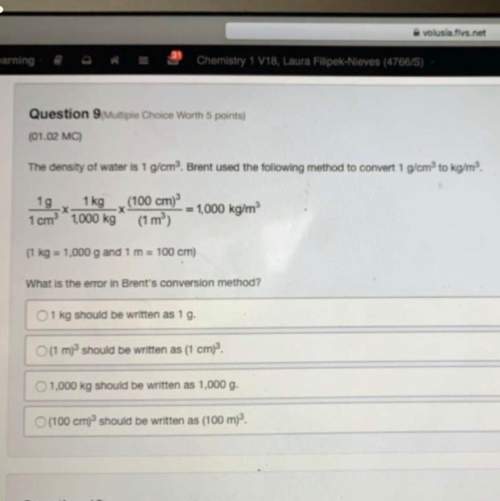
Fe2O3 + CO → Fe + CO2

NEED ANSWER NOW! 40 POINTS
Read the chemical equation.
Fe2O3 + CO → Fe + CO2
if 1.8 moles of Fe2O3 react with 2.7 moles of CO, how many moles of each product are formed?
5.4 moles Fe and 1.8 moles CO2
2.7 moles Fe and 0.9 moles CO2
3.6 moles Fe and 5.4 moles CO2
1.8 moles Fe and 2.7 moles CO2

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1


Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
You know the right answer?
NEED ANSWER NOW! 40 POINTS
Read the chemical equation.
Fe2O3 + CO → Fe + CO2
Fe2O3 + CO → Fe + CO2
Questions

Spanish, 13.10.2020 18:01


History, 13.10.2020 18:01

Mathematics, 13.10.2020 18:01


Biology, 13.10.2020 18:01



Mathematics, 13.10.2020 18:01


Mathematics, 13.10.2020 18:01

Mathematics, 13.10.2020 18:01


Social Studies, 13.10.2020 18:01


English, 13.10.2020 18:01



Computers and Technology, 13.10.2020 18:01

Biology, 13.10.2020 18:01