
Chemistry, 25.01.2021 21:10 shavonfriend27
A certain molecular compound has a solubility in acetonitrile of at . Calculate the greatest mass of that could be dissolved in of acetonitrile at this temperature. You may assume the volume of the solution doesn't change as the is dissolved. Be sure your answer has the correct unit symbol and number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1
You know the right answer?
A certain molecular compound has a solubility in acetonitrile of at . Calculate the greatest mass of...
Questions










Computers and Technology, 03.04.2020 16:42

Mathematics, 03.04.2020 16:42




Computers and Technology, 03.04.2020 16:43





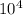 mL
mL

