
Chemistry, 29.01.2021 18:00 mmsomefood85
PLEASE HELP!!
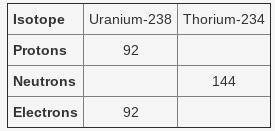
Uranium is the element used in nuclear power plants. It naturally emits particles from its nucleus. This releases energy that heats water to produce steam. The steam turns a turbine that produces electricity. The table below shows uranium-238 and thorium-234, the isotope that forms when the nucleus of uranium-238 emits a particle.
Design a test to determine whether thorium-234 also emits particles. First, explain how Rutherford’s experiment measured positive particles aimed at the gold foil. Then, explain a test that might detect whether particles are emitted from thorium-234. Last, explain why it would be more difficult to detect the release of neutrons than protons.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
How do scientists think that gravity affected the formation of our solar system?
Answers: 1

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
You know the right answer?
PLEASE HELP!!
Uranium is the element used in nuclear power plants. It naturally emits particles fro...
Questions







English, 10.01.2020 22:31

Mathematics, 10.01.2020 22:31

Computers and Technology, 10.01.2020 22:31

History, 10.01.2020 22:31

Mathematics, 10.01.2020 22:31

English, 10.01.2020 22:31





Mathematics, 10.01.2020 22:31


Arts, 10.01.2020 22:31



