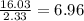Chemistry, 26.09.2019 05:30 mercymain1014
In carbon monoxide there is always 1.33 g of oxygen for every 1g carbon . how many grams of carbon are in a 16.03 g sample of carbon monoxide

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 21.06.2019 22:00
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
You know the right answer?
In carbon monoxide there is always 1.33 g of oxygen for every 1g carbon . how many grams of carbon a...
Questions

Mathematics, 29.06.2019 07:00



History, 29.06.2019 07:00


Computers and Technology, 29.06.2019 07:00



Spanish, 29.06.2019 07:00

History, 29.06.2019 07:00

History, 29.06.2019 07:00


History, 29.06.2019 07:00



Health, 29.06.2019 07:00

Mathematics, 29.06.2019 07:00