Chemistry, 22.02.2021 19:50 villarrealc1987
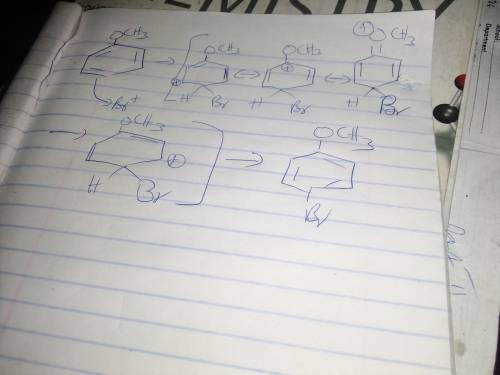
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Groups, already present on the benzene ring, that direct ortho/para further stabilize this intermediate by participating in the resonance delocalization of the positive charge. Assume that the following group is present on a benzene ring at position 1 and that you are brominating the ring at positon 4. In the box below draw the structure of the resonance contributor that shows this group actively participating in the charge delocalization. -OCH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which actions would increase the rate at salt dissolves in water? stir the water? crush the salt? use less water? heat the water? cool the salt
Answers: 3

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
You know the right answer?
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Gr...
Questions

Business, 16.10.2019 03:30

Physics, 16.10.2019 03:30

Mathematics, 16.10.2019 03:30

Chemistry, 16.10.2019 03:30

Health, 16.10.2019 03:30

Computers and Technology, 16.10.2019 03:30


History, 16.10.2019 03:30


Mathematics, 16.10.2019 03:30


Mathematics, 16.10.2019 03:30

Mathematics, 16.10.2019 03:30


Mathematics, 16.10.2019 03:30

English, 16.10.2019 03:30




Computers and Technology, 16.10.2019 03:30