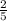Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 02:30
Which words or phrases identify layers of groundwater? check all that apply. water table kettle lake saturation zone underground lake sinkhole will give brainiest, answer quickly.
Answers: 1

Chemistry, 23.06.2019 03:00
Select the correct answer. wax is a nonpolar substance. in which type of substance is it the most soluble?
Answers: 1

You know the right answer?
Sorah, Sammie, Victor, Vicki, ond Valerie are in math group. Two students are randomly picked to
be...
Questions




Chemistry, 13.05.2021 21:40



Mathematics, 13.05.2021 21:40

Mathematics, 13.05.2021 21:40






Biology, 13.05.2021 21:40

Mathematics, 13.05.2021 21:40



Mathematics, 13.05.2021 21:40

Mathematics, 13.05.2021 21:40