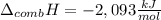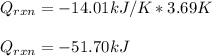Chemistry, 08.03.2021 02:50 kamster911
When 1.04g of cyclopropane was burnt in excess oxygen in a bomb calorimeter, the temperature rose by 3.69K. The total heat capacity of the calorimeter and it's contents was 14.01kJ/K. Determine the enthalpy of combustion of cyclopropane.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

You know the right answer?
When 1.04g of cyclopropane was burnt in excess oxygen in a bomb calorimeter, the temperature rose by...
Questions

Social Studies, 19.10.2019 06:00

Biology, 19.10.2019 06:00

Mathematics, 19.10.2019 06:00


English, 19.10.2019 06:10

Social Studies, 19.10.2019 06:10



Mathematics, 19.10.2019 06:10


Computers and Technology, 19.10.2019 06:10



English, 19.10.2019 06:10






History, 19.10.2019 06:10