

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

You know the right answer?
What mass of lithium phosphate would you mass to make 2.5 liter of 1.06 M lithium
phosphate solutio...
Questions


Biology, 23.06.2019 14:20












Mathematics, 23.06.2019 14:20


English, 23.06.2019 14:20




Mathematics, 23.06.2019 14:20

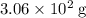 (approximately
(approximately 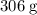 .)
.) of lithium phosphate in
of lithium phosphate in 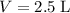 of this
of this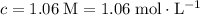 lithium phosphate solution.
lithium phosphate solution.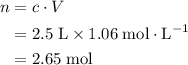 .
.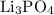 .
.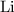 ,
, 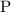 ,and
,and  on a modern periodic table:
on a modern periodic table: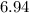 .
.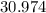 .
.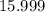 .
.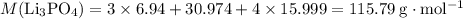 .
.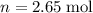 of
of  .
.

