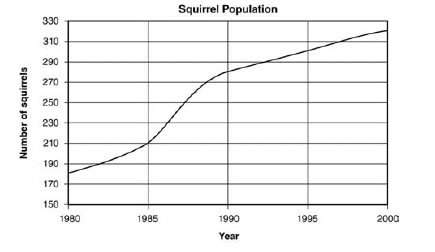
The graph below shows the size of a squirrel population over 20 years.
the trend displayed on...


Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2


Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
Questions

Mathematics, 16.01.2021 07:50

English, 16.01.2021 07:50

History, 16.01.2021 07:50

Mathematics, 16.01.2021 07:50

History, 16.01.2021 07:50

Mathematics, 16.01.2021 07:50



Spanish, 16.01.2021 07:50

Mathematics, 16.01.2021 07:50

Mathematics, 16.01.2021 07:50

Computers and Technology, 16.01.2021 07:50


Mathematics, 16.01.2021 07:50

Arts, 16.01.2021 07:50

Mathematics, 16.01.2021 07:50

Social Studies, 16.01.2021 07:50


Mathematics, 16.01.2021 07:50