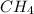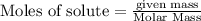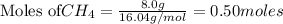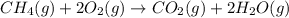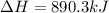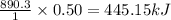Chemistry, 21.03.2021 09:50 trinityscute81
3. Consider the reaction of methane, CH4, burning in the presence of oxygen at constant pressure. Given the following equation, how much heat could be obtained by the combustion of 8.0 grams CH4? Given the thermochemical equation: CH4(g)+2O2→CO2(g)+2H2O(l); ΔH0=890.3kJ;

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2
You know the right answer?
3. Consider the reaction of methane, CH4, burning in the presence of oxygen at constant pressure. Gi...
Questions

Business, 15.06.2021 09:10



Chemistry, 15.06.2021 09:10

English, 15.06.2021 09:10


Mathematics, 15.06.2021 09:10




World Languages, 15.06.2021 09:10

Mathematics, 15.06.2021 09:10



Mathematics, 15.06.2021 09:10


Mathematics, 15.06.2021 09:10

Geography, 15.06.2021 09:10

English, 15.06.2021 09:10

Social Studies, 15.06.2021 09:10