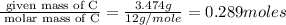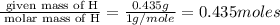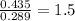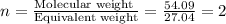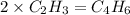Chemistry, 13.04.2021 02:00 staz13wiggins
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams of CO2 and 3.913 grams of H2O were produced. In a separate experiment, the molar mass of the compound was found to be 54.09 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2


Chemistry, 23.06.2019 02:40
How can a mixture of salt water be separated into salt and water
Answers: 1

Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
You know the right answer?
When 3.915 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 12.74 grams...
Questions



Mathematics, 20.02.2020 00:58




Mathematics, 20.02.2020 00:59





Mathematics, 20.02.2020 00:59


Biology, 20.02.2020 00:59



Advanced Placement (AP), 20.02.2020 00:59

Health, 20.02.2020 00:59

Social Studies, 20.02.2020 00:59

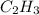 and
and  respectivley.
respectivley.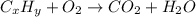
 = 12.74 g
= 12.74 g
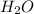 = 3.913 g
= 3.913 g
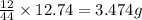 of carbon will be contained.
of carbon will be contained.
 of hydrogen will be contained.
of hydrogen will be contained.