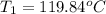Apiece of glass with a mass of 32.50 g specific heat of 0.840 j/g*°c and an initial temperature of 115 °c was dropped into a calorimeter containing 57 g of water (specific heat 4.184 j/g*°c). the final temperature of the glass and water in the calorimeter was 119.2 °c. what was the initial temperature of the water?
39.84°c
79.68°c
119.84°c
139.68°c

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 19:30
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Apiece of glass with a mass of 32.50 g specific heat of 0.840 j/g*°c and an initial temperature of 1...
Questions

Physics, 02.08.2019 09:30

History, 02.08.2019 09:30

History, 02.08.2019 09:30

History, 02.08.2019 09:30








History, 02.08.2019 09:30

History, 02.08.2019 09:30

History, 02.08.2019 09:30






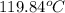
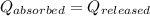
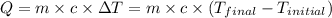
![m_1\times c_g\times (T_{final}-T_2)=-[m_2\times c_w\times (T_{final}-T_1)]](/tpl/images/0305/5772/96ec8.png) .................(1)
.................(1)
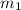 = mass of glass = 32.50 g
= mass of glass = 32.50 g
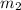 = mass of water = 57 g
= mass of water = 57 g
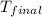 = final temperature of water and glass =
= final temperature of water and glass = 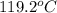
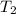 = initial temperature of water = ?
= initial temperature of water = ?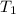 = initial temperature glass =
= initial temperature glass = 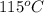
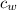 = specific heat of water =
= specific heat of water = 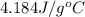
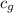 = specific heat of glass =
= specific heat of glass = 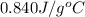
![(32.50g)\times (0.840J/g^oC)\times (119.2^oC-115^oC)=-[(57g)\times (4.184J/g^oC)\times (119.2^oC-T_1)]](/tpl/images/0305/5772/c0a00.png)