19
Gold reacts with the elements in Group 7 of the periodic table.
0.175 g of gold reacts wit...

Chemistry, 24.06.2021 14:40 jsharma57p7enrw
19
Gold reacts with the elements in Group 7 of the periodic table.
0.175 g of gold reacts with chlorine.
The equation for the reaction is:
2 Au + 3 Cl2 - 2 AuCla
Calculate the mass of chlorine needed to react with 0.175 g of gold.
Give your answer in mg
Relative atomic masses (Ar): Cl = 35.5 Au = 197

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 08:30
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1

Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1
You know the right answer?
Questions








History, 25.11.2021 08:20



Business, 25.11.2021 08:20


English, 25.11.2021 08:20


Chemistry, 25.11.2021 08:20

Mathematics, 25.11.2021 08:20



Mathematics, 25.11.2021 08:20

Mathematics, 25.11.2021 08:20

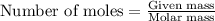 ......(1)
......(1)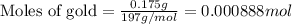
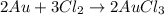
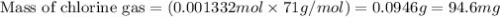 (Conversion factor: 1 g = 1000 mg)
(Conversion factor: 1 g = 1000 mg)

