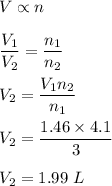Chemistry, 08.07.2021 04:40 homeschool0123
A reaction produces 3.0 mol of gas, which occupies 1.46 L. What is the volume of the product when 4.1 mol are produced at constant temperature and pressure?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
What is the correct term for living the most sustainable life you can within your current circumstances?
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
You know the right answer?
A reaction produces 3.0 mol of gas, which occupies 1.46 L. What is the volume of the product when 4....
Questions

English, 13.11.2020 20:30

Geography, 13.11.2020 20:30

History, 13.11.2020 20:30

Mathematics, 13.11.2020 20:30



Biology, 13.11.2020 20:30







Health, 13.11.2020 20:30




Engineering, 13.11.2020 20:30


Mathematics, 13.11.2020 20:30