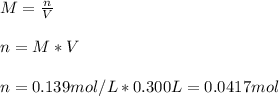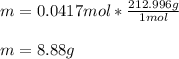Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

You know the right answer?
you ned to make an aqeous solution of .139M aluminum nitrate for an experiment in a lab, using 300....
Questions



Advanced Placement (AP), 08.10.2019 14:00



Physics, 08.10.2019 14:10

Spanish, 08.10.2019 14:10


Mathematics, 08.10.2019 14:10


Mathematics, 08.10.2019 14:10

Mathematics, 08.10.2019 14:10




History, 08.10.2019 14:10

Biology, 08.10.2019 14:10



Mathematics, 08.10.2019 14:10