1 point
3. According to Table 3, how do the different types of 5-carbon molecules
differ wit...

Chemistry, 29.09.2021 19:20 nonjabulomabaso6850
1 point
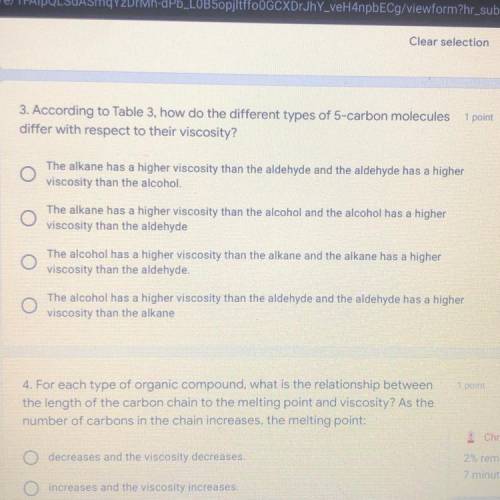
3. According to Table 3, how do the different types of 5-carbon molecules
differ with respect to their viscosity?
The alkane has a higher viscosity than the aldehyde and the aldehyde has a higher
viscosity than the alcohol.
The alkane has a higher viscosity than the alcohol and the alcohol has a higher
viscosity than the aldehyde
The alcohol has a higher viscosity than the alkane and the alkane has a higher
viscosity than the aldehyde.
The alcohol has a higher viscosity than the aldehyde and the aldehyde has a higher
viscosity than the alkane

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2


You know the right answer?
Questions



Business, 21.05.2021 17:30

History, 21.05.2021 17:30








Computers and Technology, 21.05.2021 17:30



Advanced Placement (AP), 21.05.2021 17:30

Mathematics, 21.05.2021 17:30

Mathematics, 21.05.2021 17:30

Mathematics, 21.05.2021 17:30

Mathematics, 21.05.2021 17:30



