
Chemistry, 07.12.2021 05:50 alexmiranda00
PLEASE HELP WILL GIVE BRAINLISET AND POINTS!! For an experiment, a biology student makes 1 liter of a solution containing the substances shown in the table below. Using the given information, fill in the missing parts of the table for each substance by (a) providing the formula weight and (b) calculating either the mass the student needs to weigh out or the number of moles that the student has, based on the amount weighed out. For each calculation, show your work. Be sure to include the formula mass of water for CaCl2 • 2H2O and Na(CH3)2 AsO2 • 3H2O.
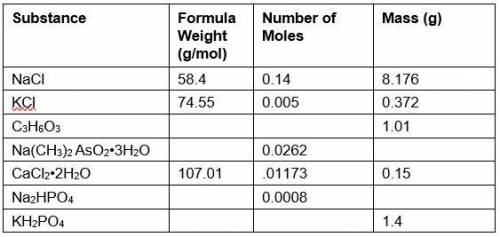

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 23.06.2019 03:10
Which is true according to the law of conservation of energy
Answers: 1

Chemistry, 23.06.2019 12:00
372 ml is the volume of aluminum, density is 2.70 g/ml what is the mass in grams
Answers: 1
You know the right answer?
PLEASE HELP WILL GIVE BRAINLISET AND POINTS!!
For an experiment, a biology student makes 1 liter o...
Questions


Biology, 28.07.2019 01:00

English, 28.07.2019 01:00


Biology, 28.07.2019 01:00




Mathematics, 28.07.2019 01:00

Mathematics, 28.07.2019 01:00




Mathematics, 28.07.2019 01:00

Biology, 28.07.2019 01:00



Biology, 28.07.2019 01:00

Mathematics, 28.07.2019 01:00



