
Chemistry, 28.07.2019 14:00 vernelljordon
For the nitrogen fixation reaction, 3h2(g) + n2(g) 2nh3(g), kc = 6.0 × 10–2 at 500°c. if 0.250 m h2 and 0.050 m nh3 are present at equilibrium, what is the equilibrium concentration of n2? a. 3.3 m b. 2.7 m c. 0.20 m d. 0.083 m e. 0.058 m

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1
You know the right answer?
For the nitrogen fixation reaction, 3h2(g) + n2(g) 2nh3(g), kc = 6.0 × 10–2 at 500°c. if 0.250 m h2...
Questions


Law, 23.10.2020 05:01


Advanced Placement (AP), 23.10.2020 05:01

Spanish, 23.10.2020 05:01


Mathematics, 23.10.2020 05:01



Geography, 23.10.2020 05:01


Mathematics, 23.10.2020 05:01


Mathematics, 23.10.2020 05:01

Mathematics, 23.10.2020 05:01


History, 23.10.2020 05:01



![Kc=\frac{[NH_3]^2_{eq}}{[H_2]^3_{eq}[N_2]_{eq}}](/tpl/images/0143/1009/c1580.png)
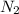 is the unique unknown, solving for it one obtains:
is the unique unknown, solving for it one obtains:![[N_2]_{eq}=\frac{[NH_3]^2_{eq}}{[H_2]^3_{eq}Kc}](/tpl/images/0143/1009/e7601.png)
![[N_2]_{eq}=\frac{(0.050M)^2}{(0.250M)^3(6.0x10^{-2})}](/tpl/images/0143/1009/c87cf.png)
![[N_2]_{eq}=2.7M](/tpl/images/0143/1009/d60cf.png)


