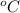Physics, 08.07.2020 06:01 saadshami2384
A gas occupies a certain volume at 27°C. At what temperature will it volume be doubled, assuming that its pressure remains constant?

Answers: 2
Another question on Physics

Physics, 22.06.2019 07:10
1. how much energy is needed to raise the temperature of 40.0 g of argon from 25c to 40c? the specific heat capacity of argon is 0.520 j/(g·k) 2a. 23.0 ml of 0.100 m hcl (standard) are added from a buret to neutralize 50.0 ml of an unknown basic solution. 2b. if the oh- produced in the previous reaction came from ca(oh)2, then what is the molarity of the ca(oh)2? 3.calculate the new freezing-point of a solution when 60.5 grams of cacl2 solute is dissolved in 0.612 kg of water. 4. what is the maximum number of moles of alcl3 that can be produced from 5.0 mol al and 6.0 mol cl2? 5. a sample of oxygen gas has a volume of 150 ml when its pressure is 0.923 atm. if the pressure is increased to 0.987 atm and the temperature remains constant, what will the new volume be? 6. nitrogen gas in a closed container at a temperature of 100.0 oc and 3.0 atm is heated to 300 oc. what is the pressure of the gas at the higher temperature?
Answers: 3

Physics, 22.06.2019 08:00
If a substance is in the gas phase, which of qualities of the gas will stay constant? a: volume b: mass c: shape d: position of particles
Answers: 2

Physics, 22.06.2019 08:30
Determine the age of a fossil if it had only one eighth of its original carbon-14 content remaining?
Answers: 3

Physics, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1
You know the right answer?
A gas occupies a certain volume at 27°C. At what temperature will it volume be doubled, assuming tha...
Questions

History, 27.07.2019 17:50










Social Studies, 27.07.2019 17:50

Biology, 27.07.2019 17:50








Chemistry, 27.07.2019 17:50