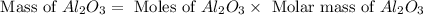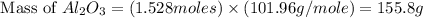Chemistry, 23.07.2019 13:20 paranoidbih
Aluminum oxide (used as an adsorbent or a catalyst for organic reactions) forms when aluminum reacts with oxygen. 4al(s) + 3o2(g) ® 2al2o3(s) [balanced] a mixture of 82.49 g of aluminum ( = 26.98 g/mol) and 117.65 g of oxygen ( = 32.00 g/mol) is allowed to react. what mass of aluminum oxide ( = 101.96 g/mol) can be formed?

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
You know the right answer?
Aluminum oxide (used as an adsorbent or a catalyst for organic reactions) forms when aluminum reacts...
Questions

Computers and Technology, 14.12.2019 10:31


Spanish, 14.12.2019 10:31



Mathematics, 14.12.2019 10:31

Health, 14.12.2019 10:31


Mathematics, 14.12.2019 10:31



Mathematics, 14.12.2019 10:31


Spanish, 14.12.2019 10:31

Mathematics, 14.12.2019 10:31





Chemistry, 14.12.2019 10:31

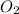 = 117.65 g
= 117.65 g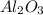 = 101.96 g/mole
= 101.96 g/mole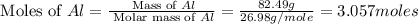
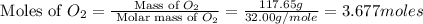
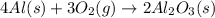
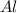 react with 3 mole of
react with 3 mole of 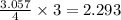 moles of
moles of 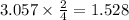 moles of
moles of