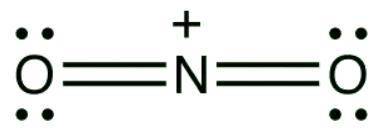A mixture of sulfuric acid and nitric acid will produce small quantities of the nitronium ion (NO2 ): The nitronium ion has a central nitrogen atom with a positive charge double bonded to two oxygen atoms on both sides. Each oxygen atom carries two lone pairs of electrons. Does the nitronium ion have any significant resonance structures

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

You know the right answer?
A mixture of sulfuric acid and nitric acid will produce small quantities of the nitronium ion (NO2 )...
Questions


Mathematics, 17.05.2021 17:50

Mathematics, 17.05.2021 17:50



Mathematics, 17.05.2021 17:50







Mathematics, 17.05.2021 17:50


Mathematics, 17.05.2021 17:50

Law, 17.05.2021 17:50

English, 17.05.2021 17:50